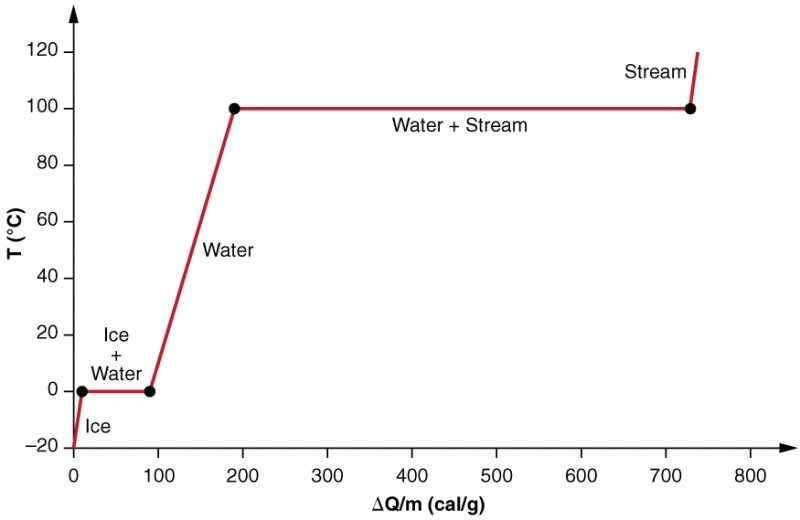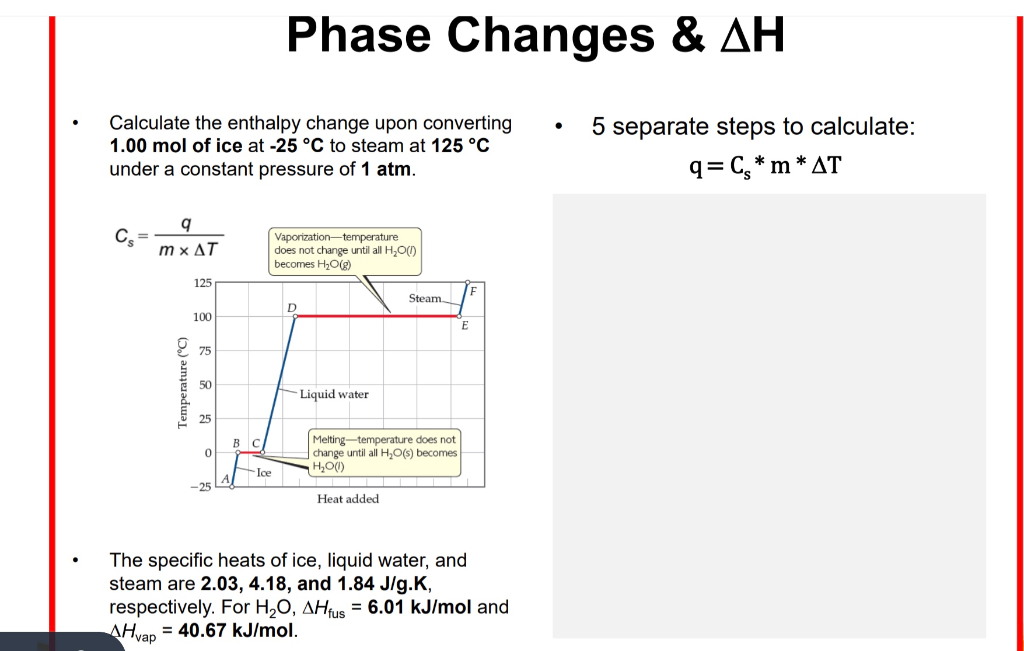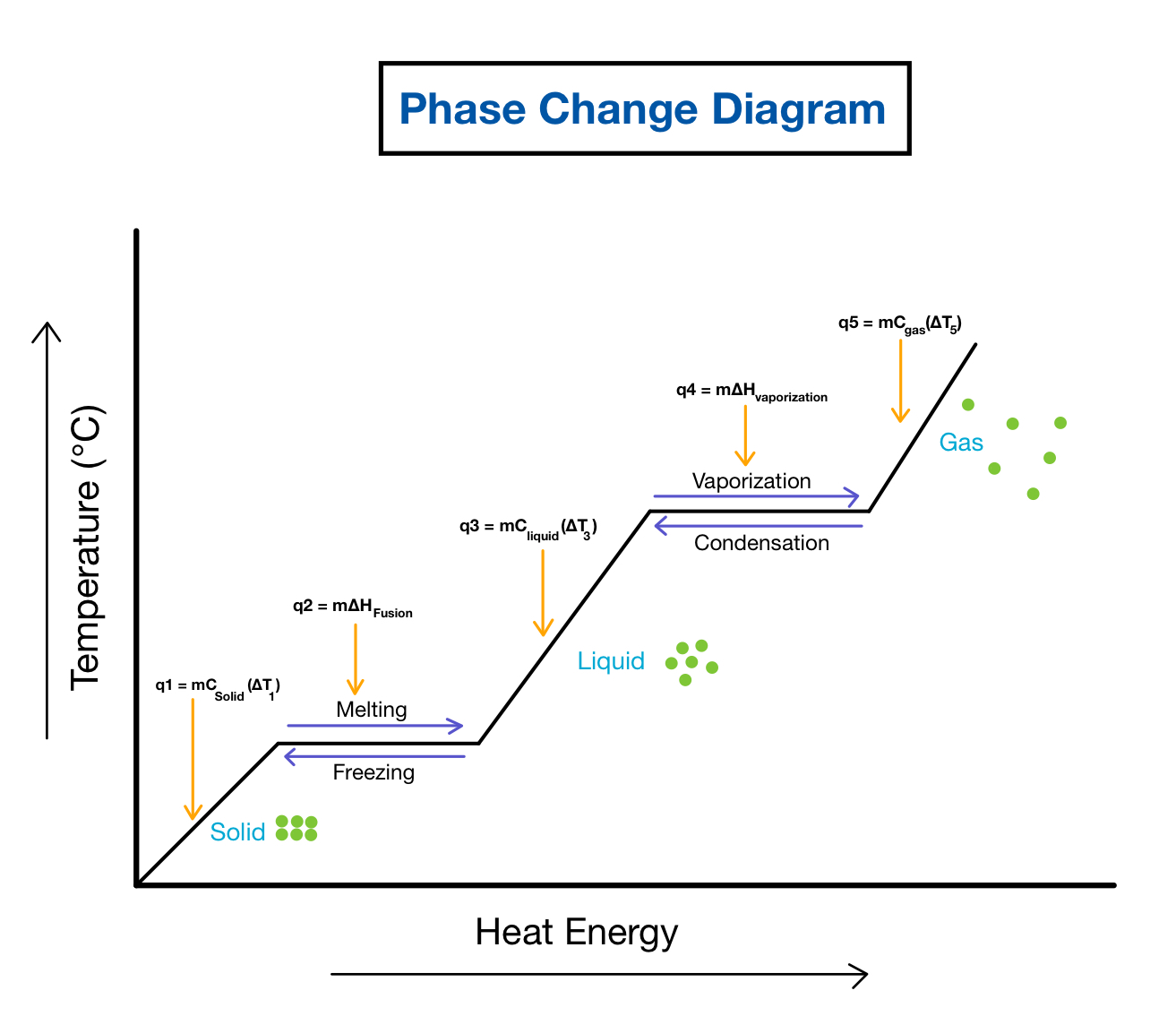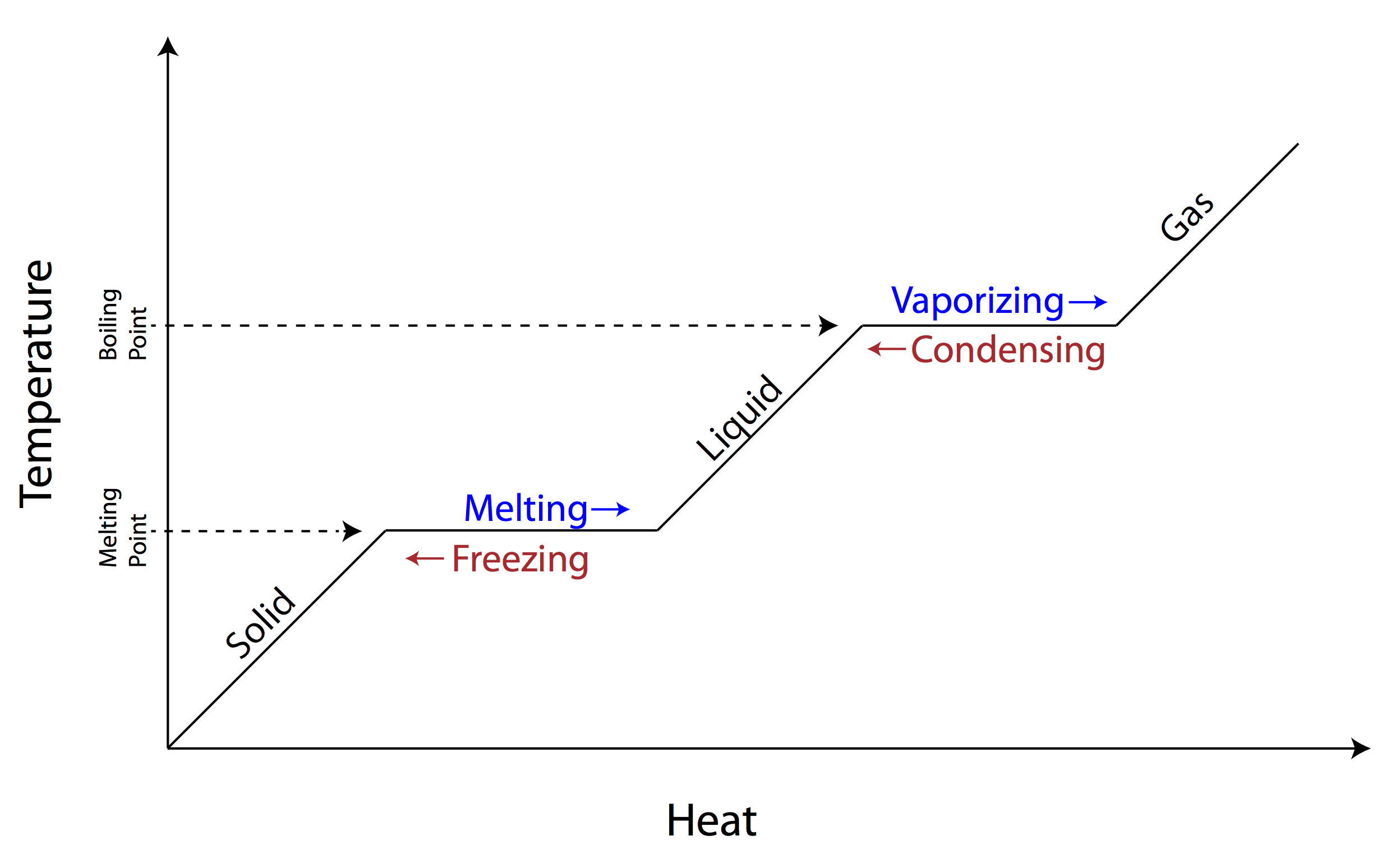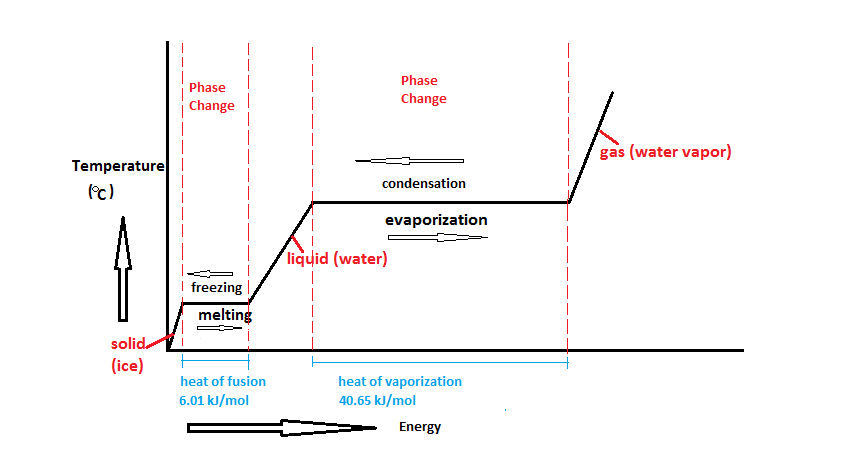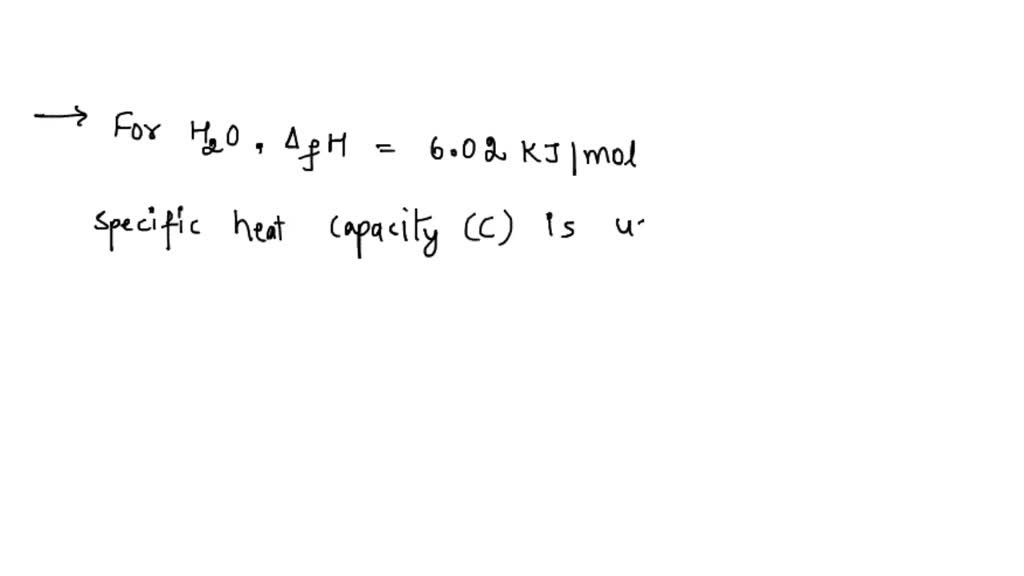
SOLVED: Heat of fusion (ΔHfus) is used for calculations involving a phase change between solid and liquid, with no temperature change. For H2O, ΔHfus=6.02 kJ/mol. Specific heat capacity (C) is used for

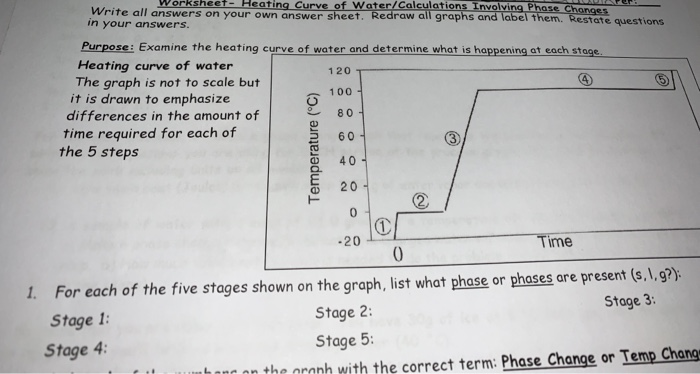
HeatingCurveofWaterWorksheet - Acc Chem Q4 HW #6.pdf - Name: Per: Worksheet- Heating Curve of Water/Calculations Involving Phase Changes Write all | Course Hero