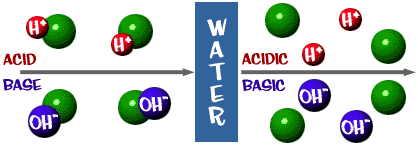
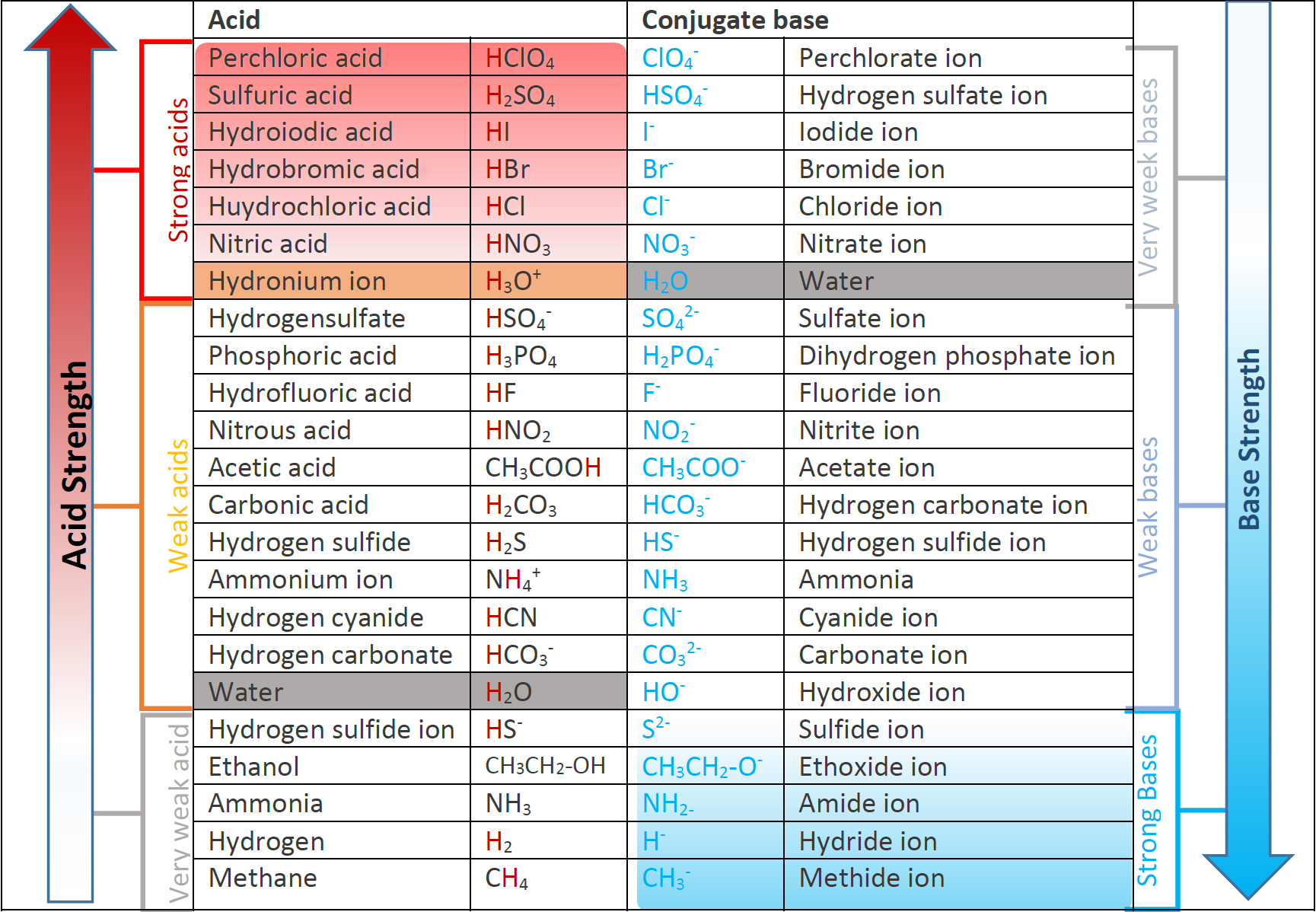
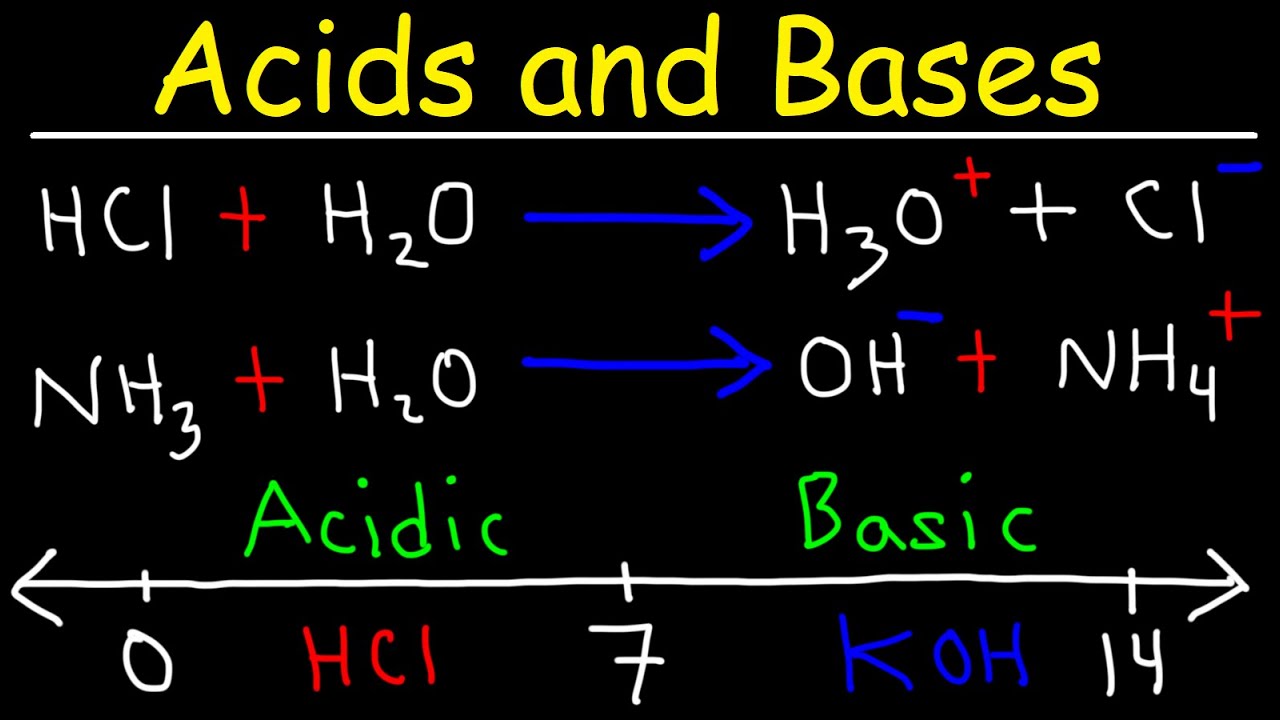
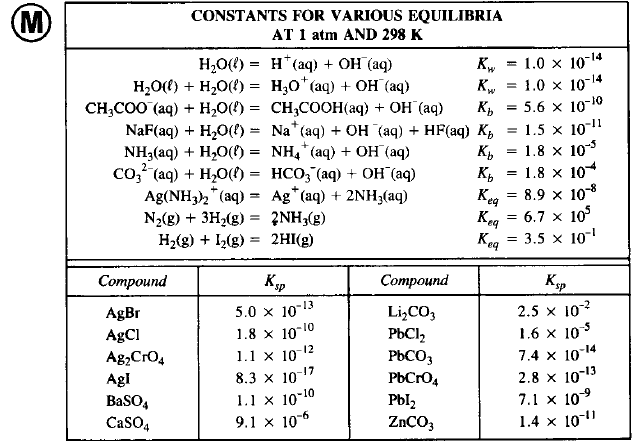
Identify the conjugate acid-base pairs for the reaction (with the acid written first). CN- + H2O = HCN + OH- |CN- / HCN |HCN / CN- |OH- / H2O |H2O / OH- | Homework.Study.com
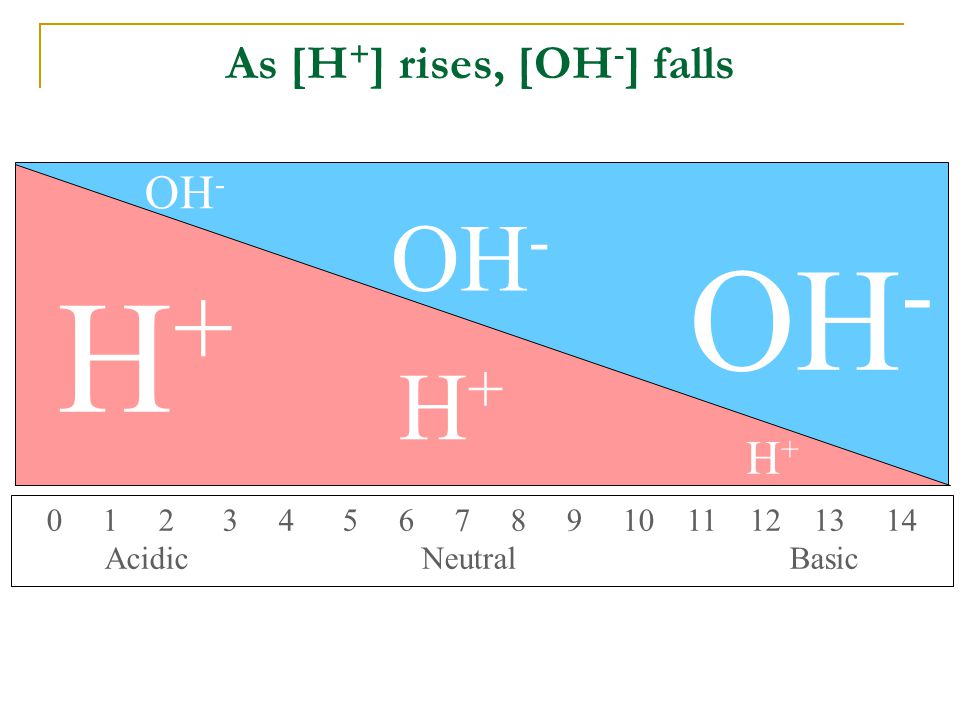
Acids, Bases, Salts and Buffers Experiment 8. #8 Acids, bases, salts and buffers Goals: Understand weak acid (base) equilibria and conjugate acid- base. - ppt download

![Calculating pH, pOH, [H+], [OH-] - Acids and Bases Calculating pH, pOH, [H+], [OH-] - Acids and Bases](http://iloveacid--basechemistry.weebly.com/uploads/2/7/8/0/27808151/2222607_orig.gif)