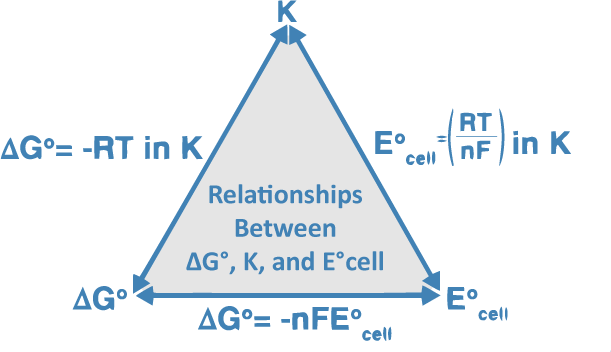
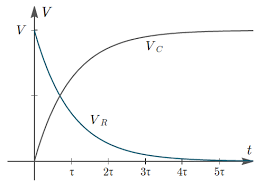
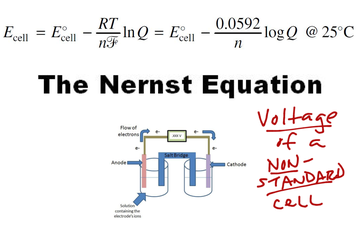
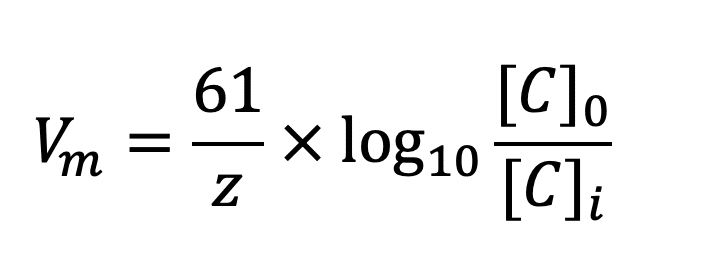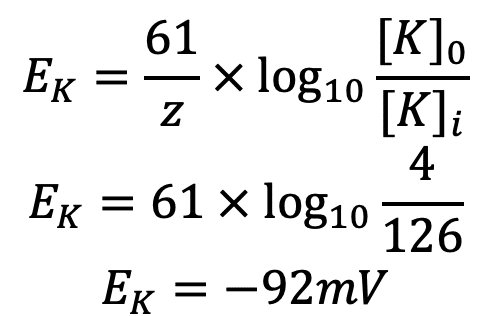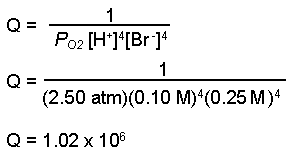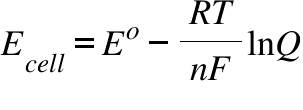Why do we use the Nernst equation in biology? What are some examples? How do we calculate NERNST with 2 different ions in a cell? - Quora
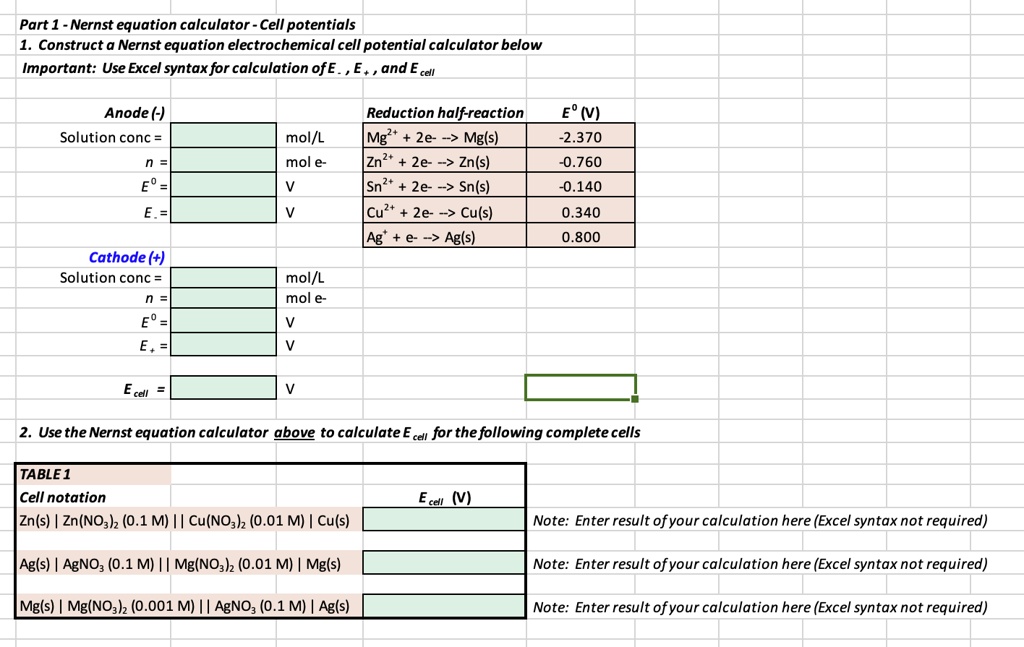
SOLVED: Part 1 Nernst equation calculator - Cell potentials Construct a Nernst equation electrochemical cell potential calculator below Important: Use Excel syntaxfor calculation ofE and E cel Anode (-) Solution conc Reduction

Membrane Potential Overview, Equation, & Functions | What is Membrane Potential? - Video & Lesson Transcript | Study.com