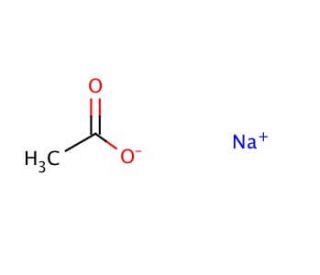
![Using sodium acetate for the synthesis of [Au(NHC)X] complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/D0DT02240C Using sodium acetate for the synthesis of [Au(NHC)X] complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/D0DT02240C](https://pubs.rsc.org/image/article/2020/DT/d0dt02240c/d0dt02240c-u1_hi-res.gif)
Using sodium acetate for the synthesis of [Au(NHC)X] complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/D0DT02240C

Growth of S. salsiraiae IM408 under different sodium acetate (NaOAc)... | Download Scientific Diagram
Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora

If sodium acetate is a weak acid and does not readily dissociate in water or completely and a strong electrolyte is defined as the oppposite how come the answer is B and

Sodium acetate hydrate, Puratronic , 99.9985% (metals basis), Thermo Scientific Chemicals | Fisher Scientific

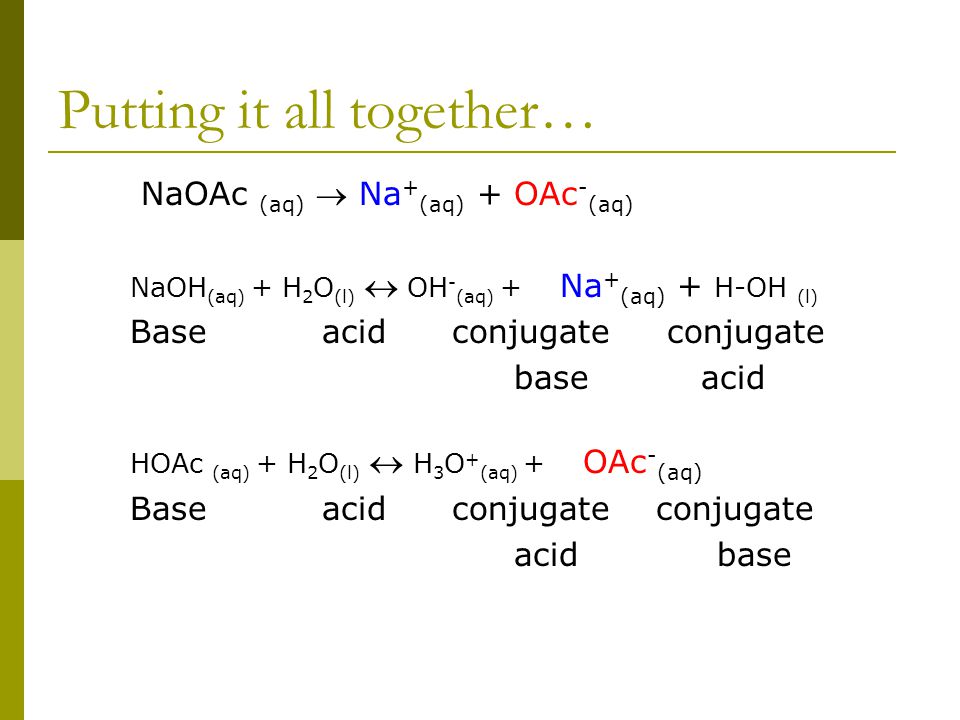
SOLVED: Sodium acetate (NaC2H3O2) is a basic salt. When sodium acetate is dissolved in water, it dissociates into its component ions. This reaction goes to completion, as indicated by the one-way arrow

Scheme 1. (a) Br2/NaOAc-buffer, r.t., overnight, 81%. (b) Pd(PPh3)4... | Download Scientific Diagram
![Sodium Acetate-promoted Oxa-Michael-Aldol [3+2] Annulation Reactions: Facile Access to the Fused Heterocycle | Bentham Science Sodium Acetate-promoted Oxa-Michael-Aldol [3+2] Annulation Reactions: Facile Access to the Fused Heterocycle | Bentham Science](https://www.eurekaselect.com/images/graphical-abstract/ccat/7/1/006.jpg)
Sodium Acetate-promoted Oxa-Michael-Aldol [3+2] Annulation Reactions: Facile Access to the Fused Heterocycle | Bentham Science
![Using sodium acetate for the synthesis of [Au(NHC)X] complexes - Dalton Transactions (RSC Publishing) Using sodium acetate for the synthesis of [Au(NHC)X] complexes - Dalton Transactions (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/D0DT02240C)
Using sodium acetate for the synthesis of [Au(NHC)X] complexes - Dalton Transactions (RSC Publishing)