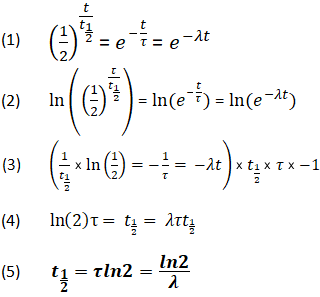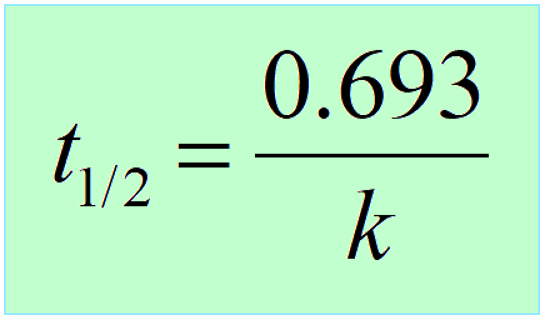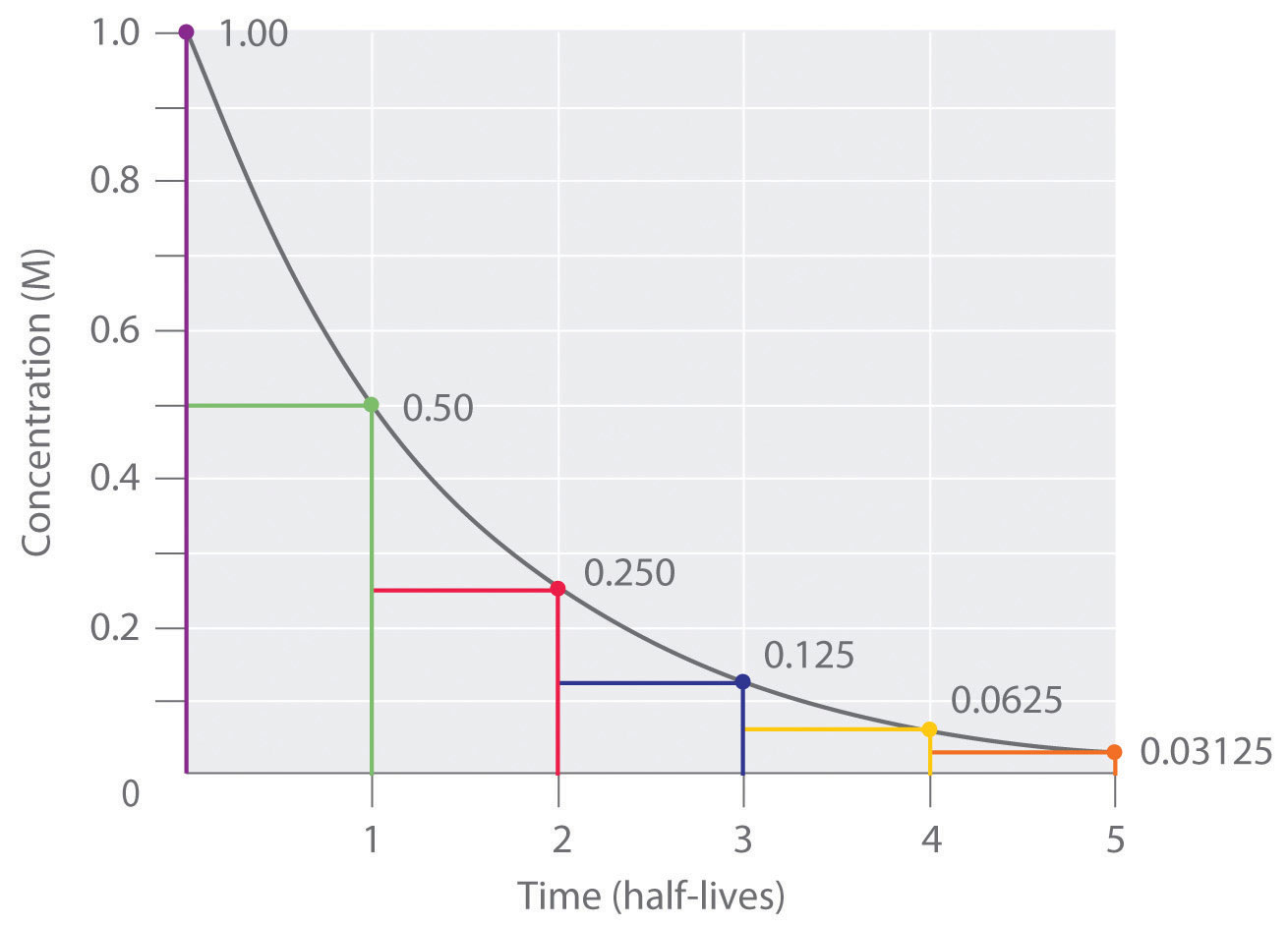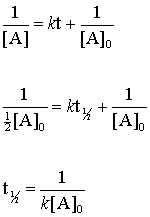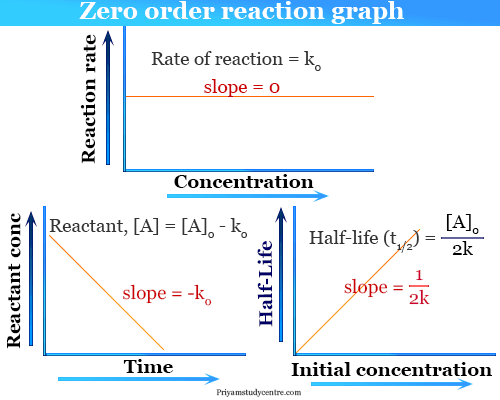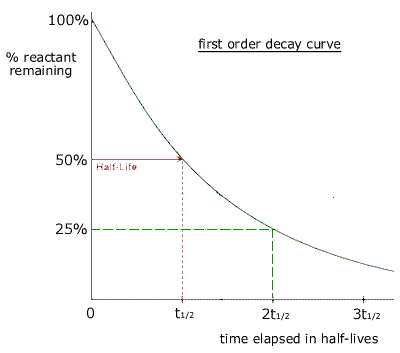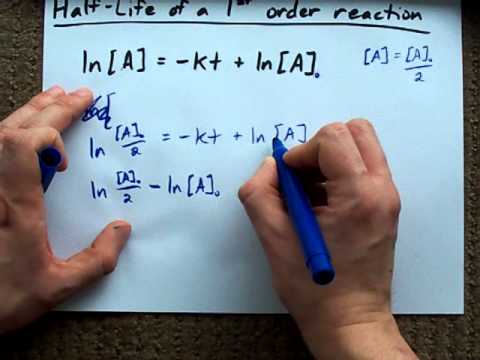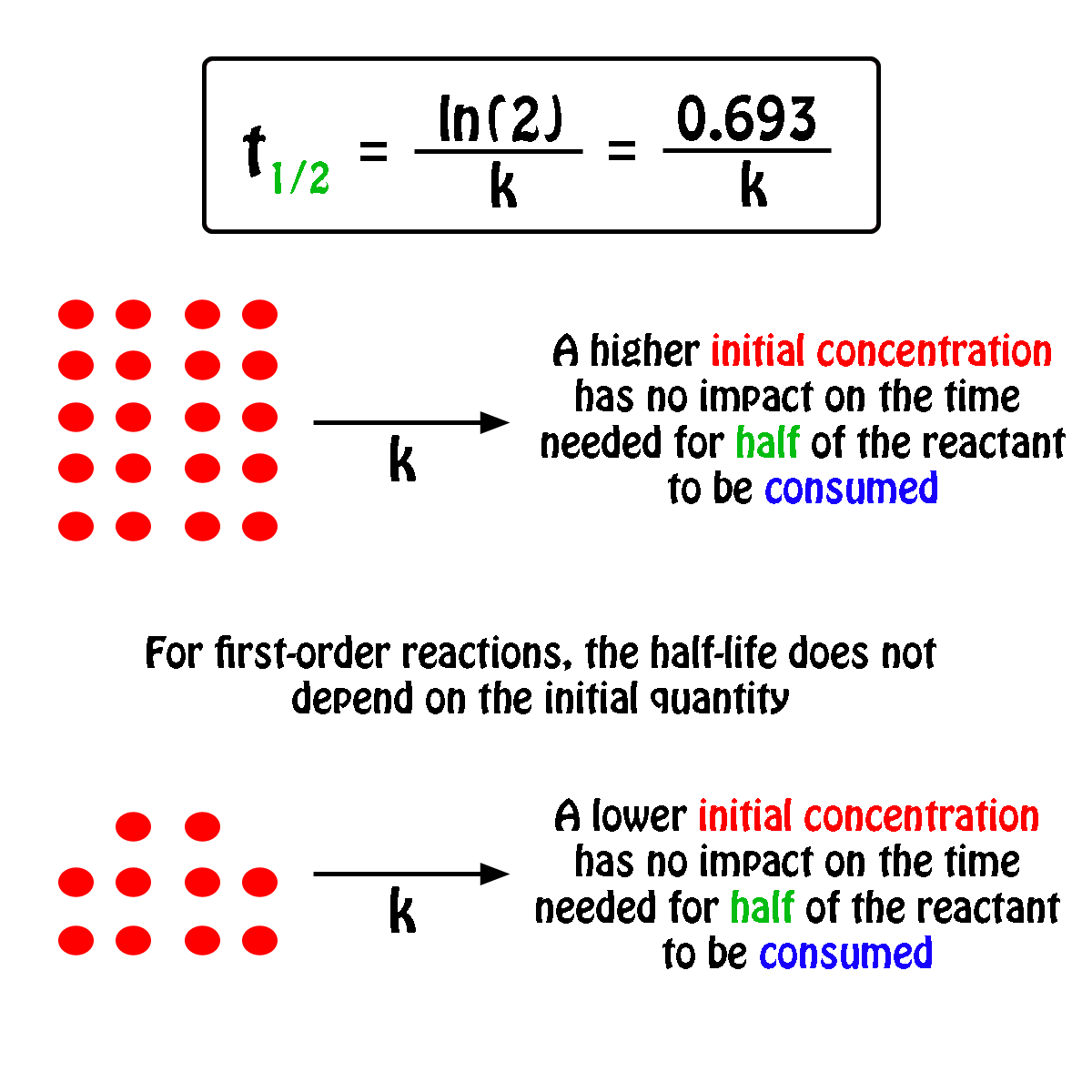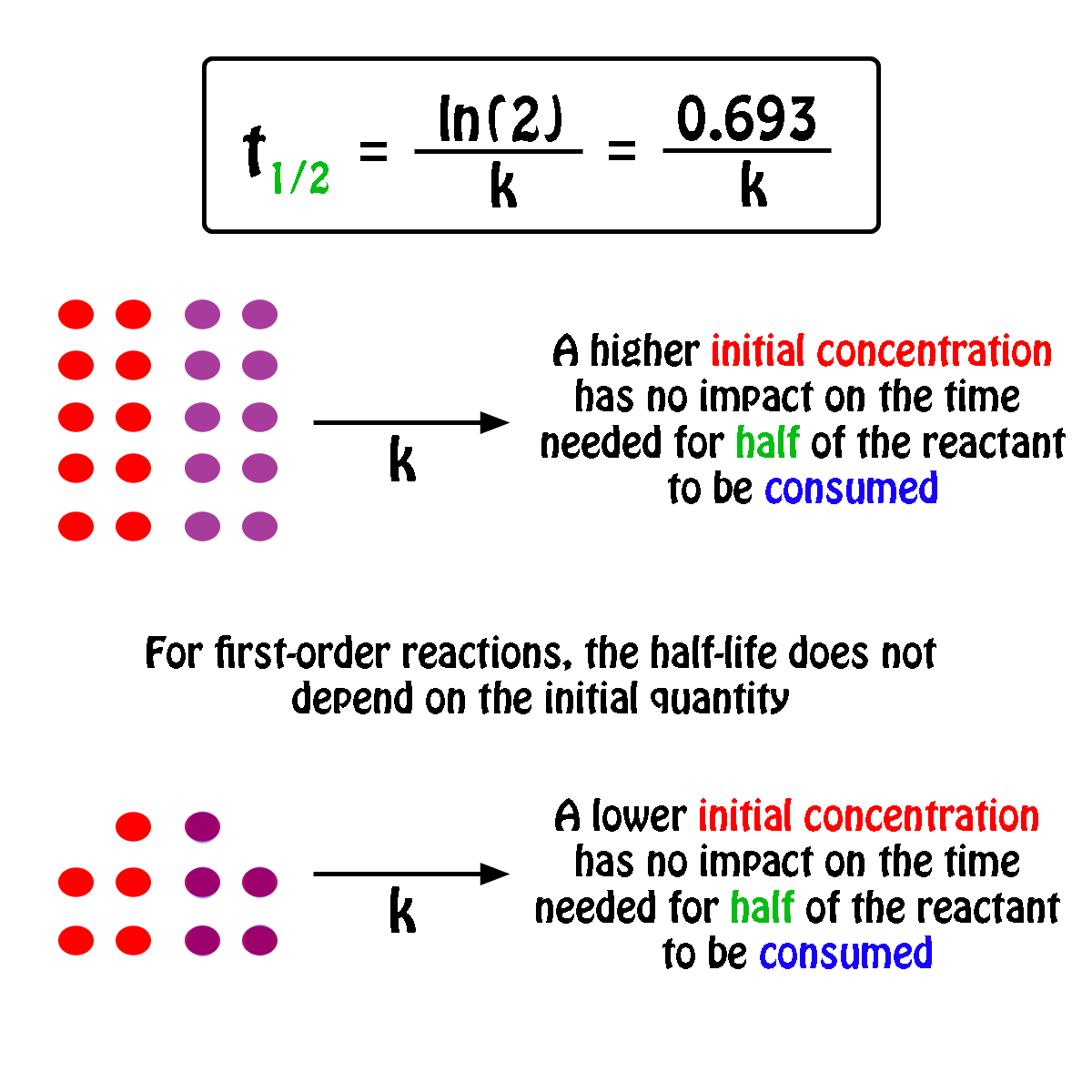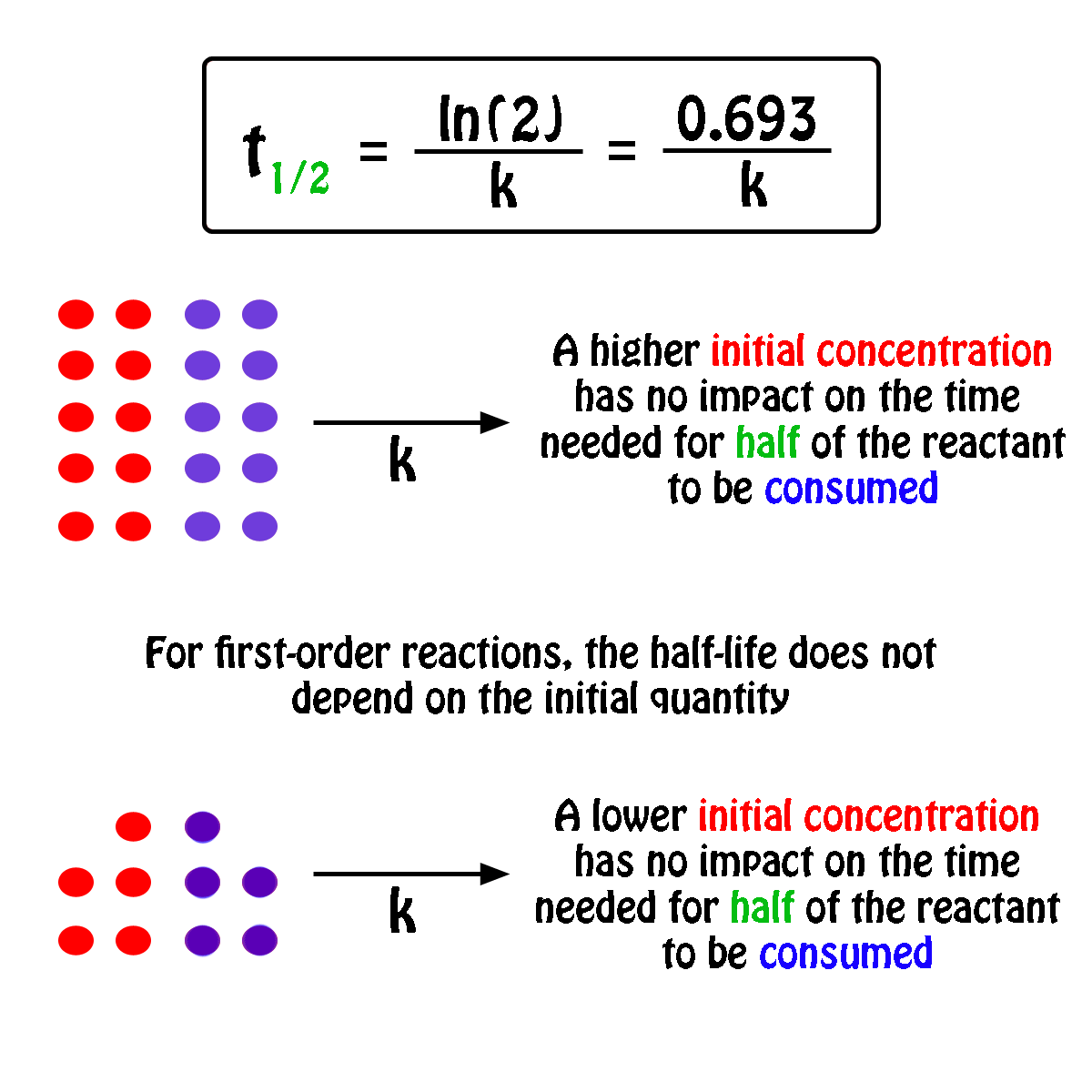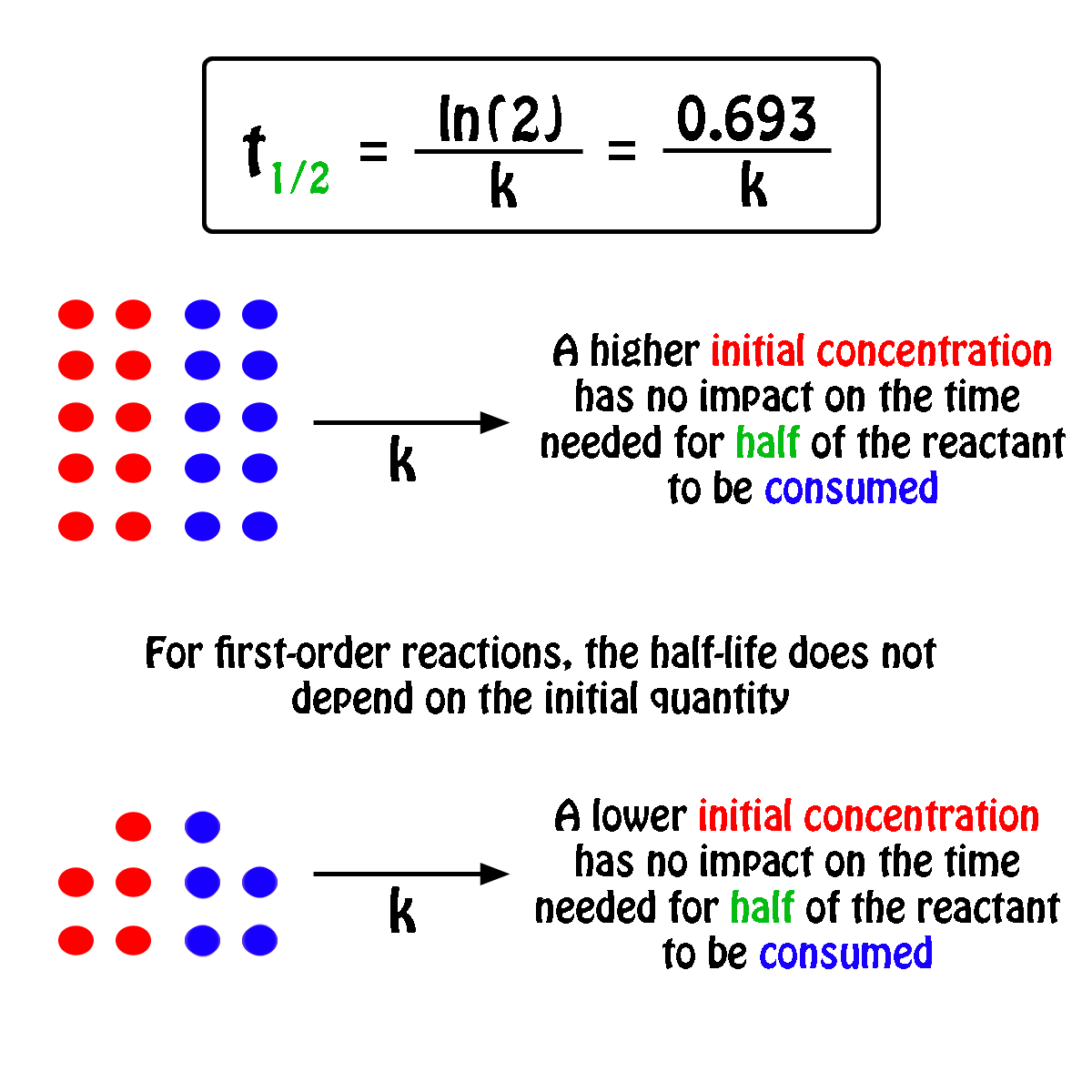
What is the half-life of a first-order reaction with a rate constant of 7.80xx10^-4 s^(-1)? | Socratic

Calculate the half life of a first order reaction from their rate constants given below:(a) 200 s^-1 ; (b) 2 min^-1 ; (c) 4 year^-1 .
The formula of half life for an n^th order reaction involving reactant A and n ≠ 1 is - Sarthaks eConnect | Largest Online Education Community