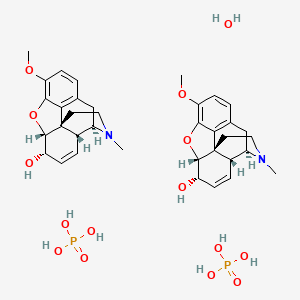
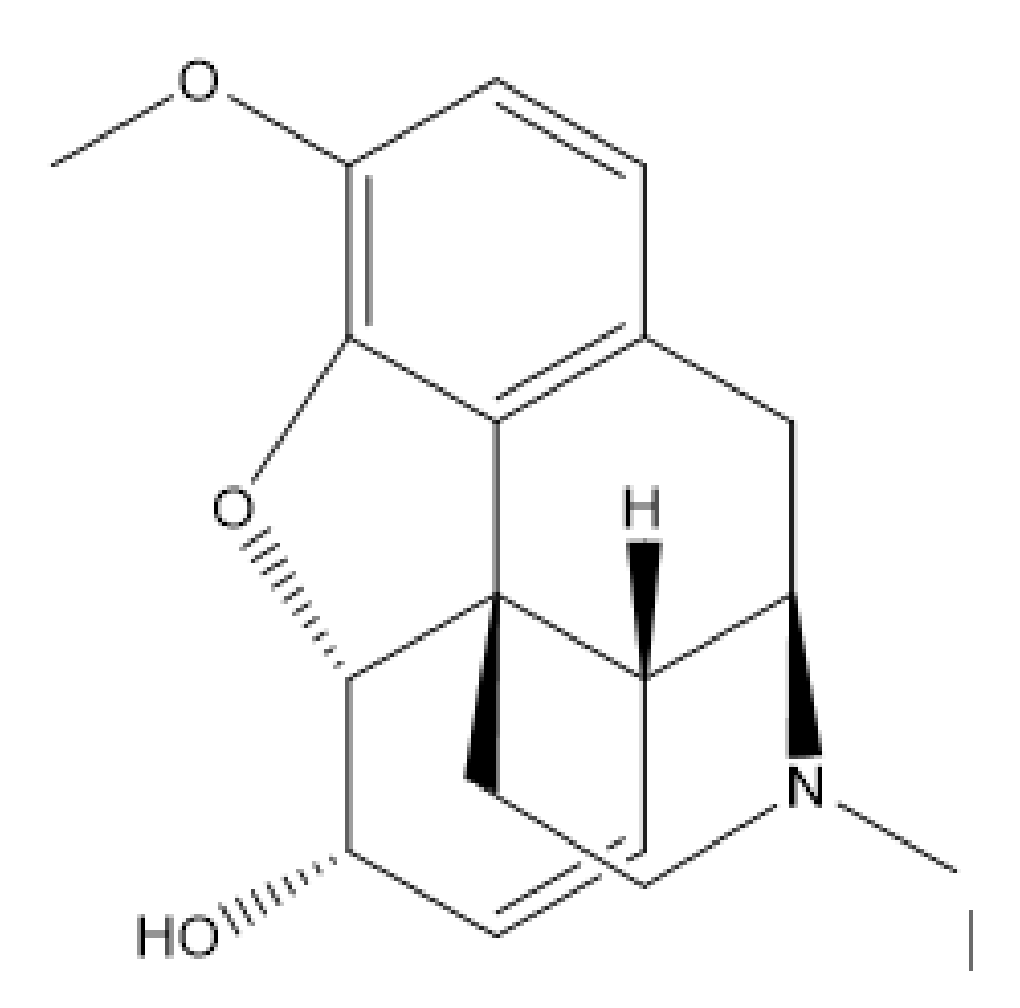
You need to prepare 24 suppositories each containing 60 mg of codeine phosphate in Hard Fat BP base - YouTube

SOLVED: The substance codeine is a weak nitrogenous base like ammonia Write net ionic equation tO show how codeine; C1sHz O,N, behaves as base In Water 1o

Norcodeine (3-O-methyl Normorphine, N-desmethyl Codeine, Normorphine 3-methyl ether, CAS Number: 467-15-2) | Cayman Chemical
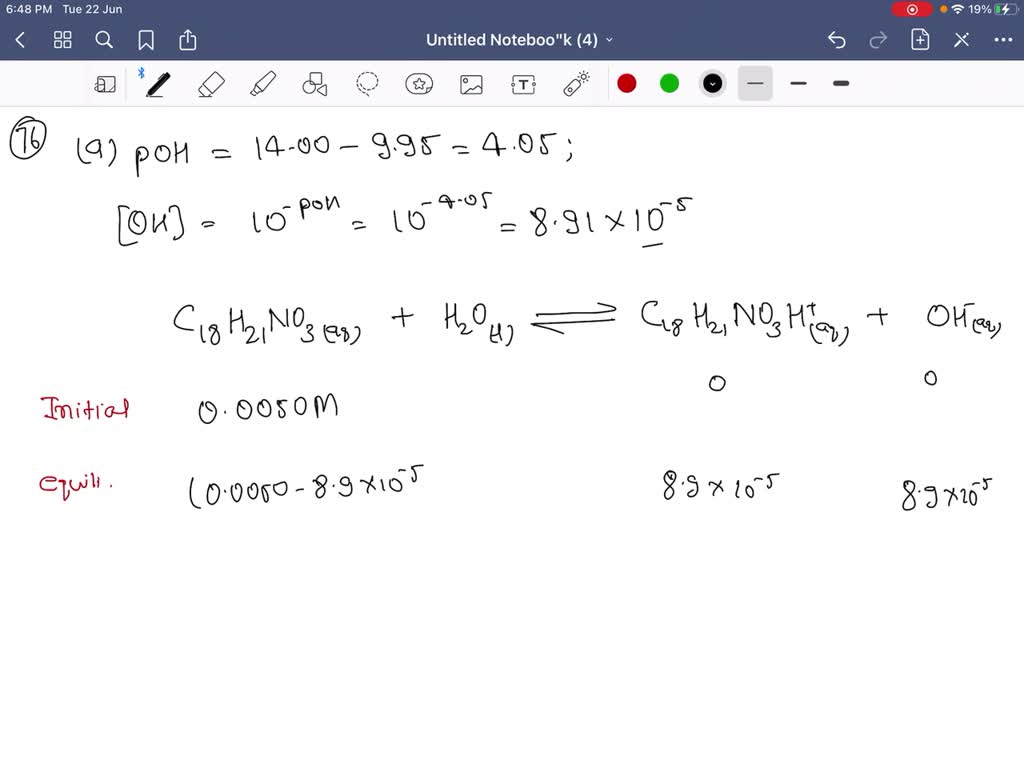
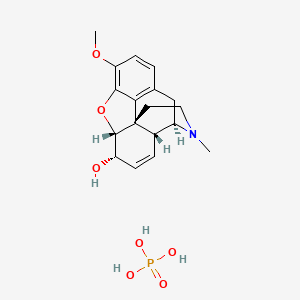
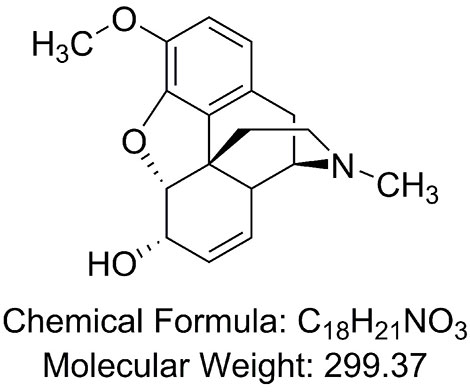
SOLVED:Codeine (C18 H21 NO3) is a weak organic base. A 5.0 ×10^-3 M solution of codeine has a pH of 9.95 . Calculate the value of Kb for this substance. What is

Codeine is added to 100 mL of water until the solution concentration is 0.800 M. What is the pH of this solution? | Homework.Study.com
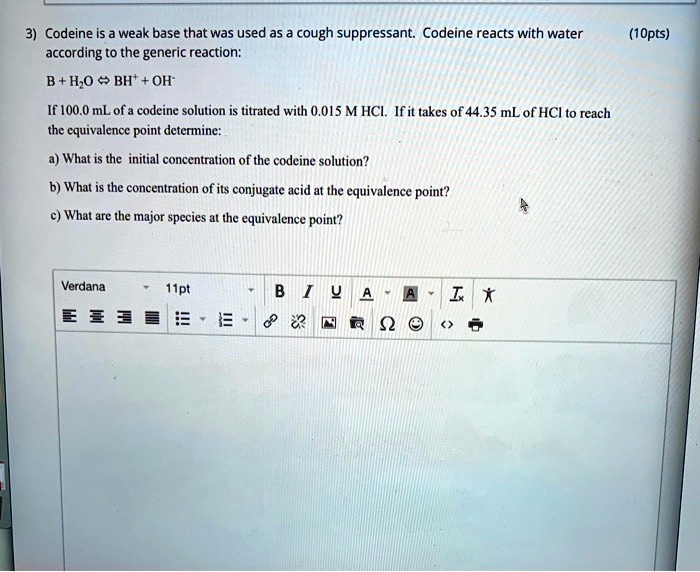
SOLVED: Codeine is weak base that was used as cough suppressant: Codeine reacts with water according to the generic reaction: H,o < BH' OH" (1Opts) If 100.0 mL of a codeine solution