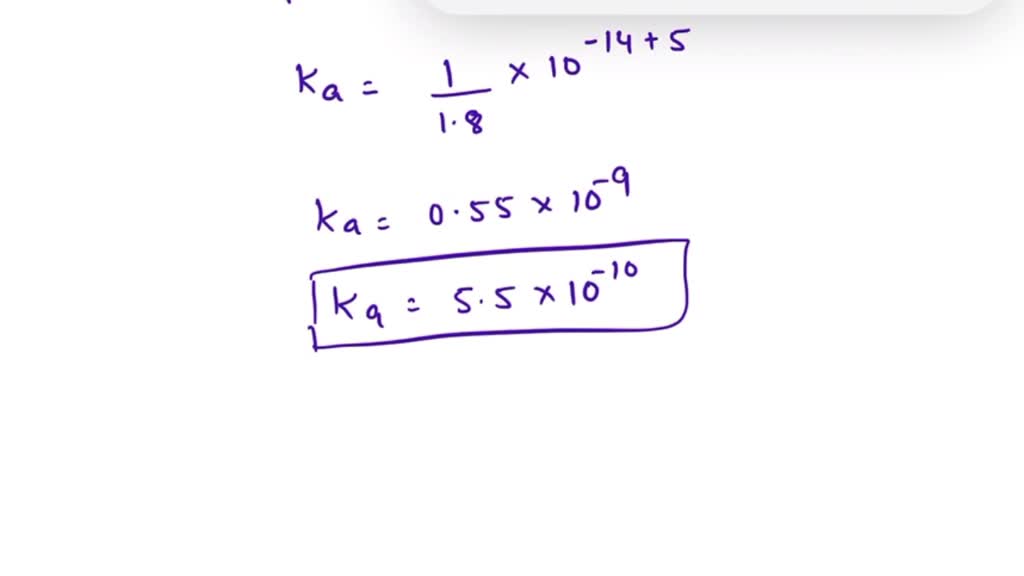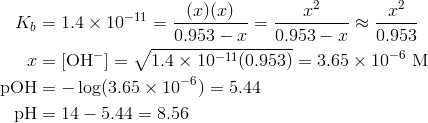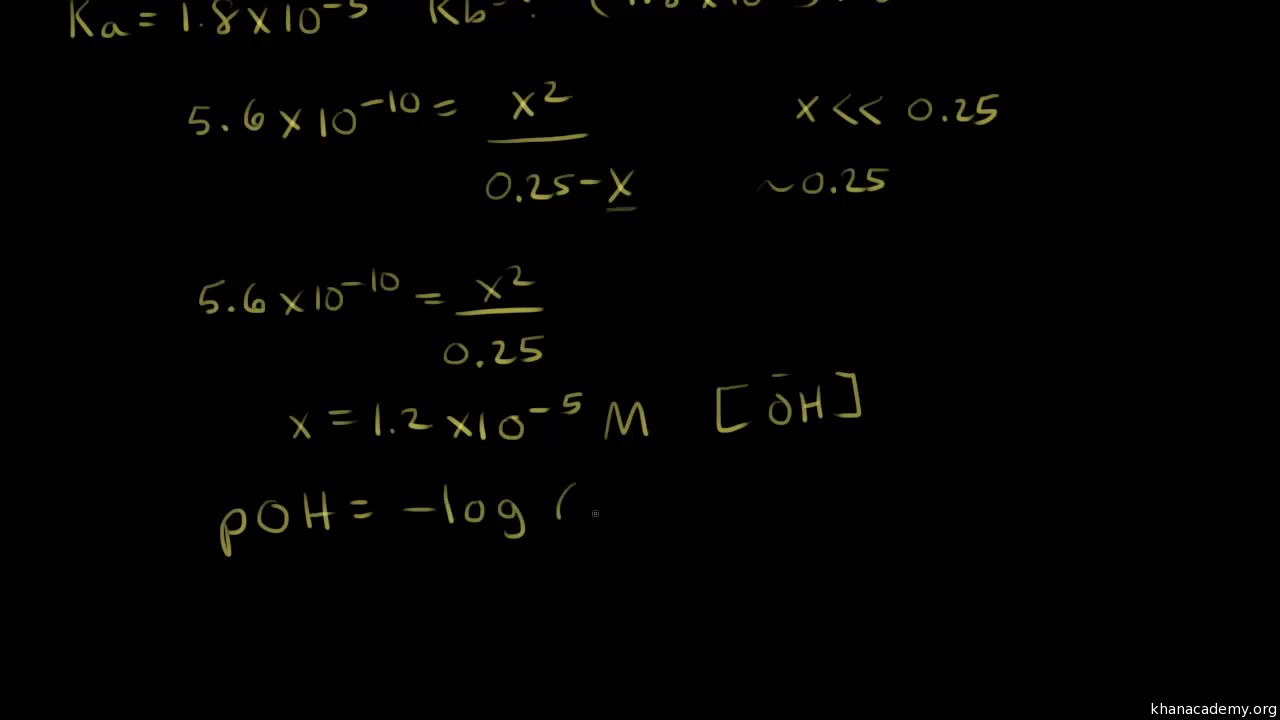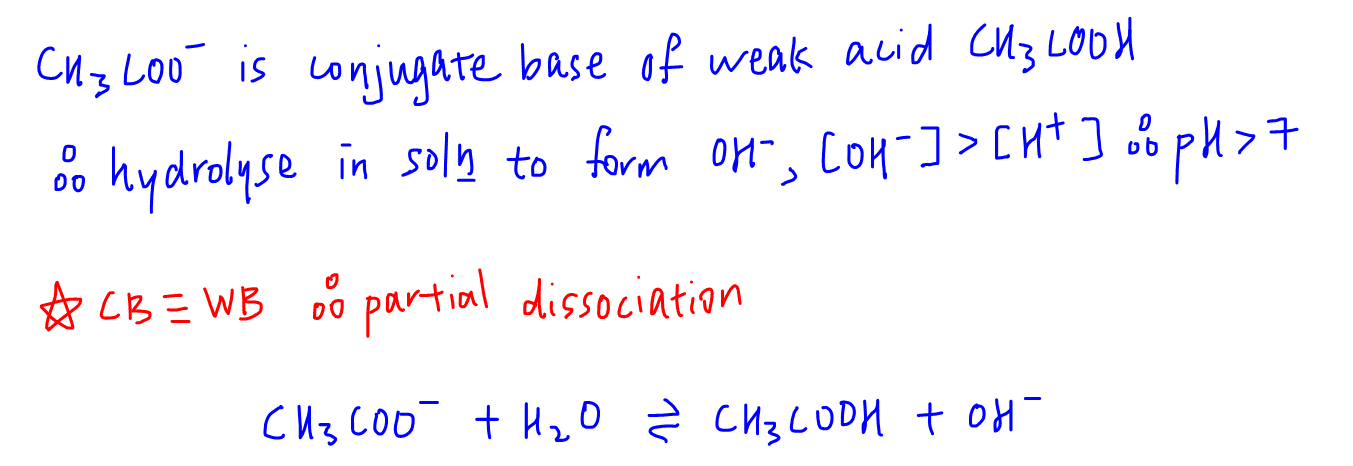![Pure water is netural in nature [H^(+)]=[OH^(-)]. When this condition is disturbed by changing the concentration of H^(+)or OH^(-), the natural solution changes to acidic {[H^(+)]gt[OH^(-)]} or basic {[H^(+)]lt[OH^(-)]}. This change occurs Pure water is netural in nature [H^(+)]=[OH^(-)]. When this condition is disturbed by changing the concentration of H^(+)or OH^(-), the natural solution changes to acidic {[H^(+)]gt[OH^(-)]} or basic {[H^(+)]lt[OH^(-)]}. This change occurs](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/644354016_web.png)
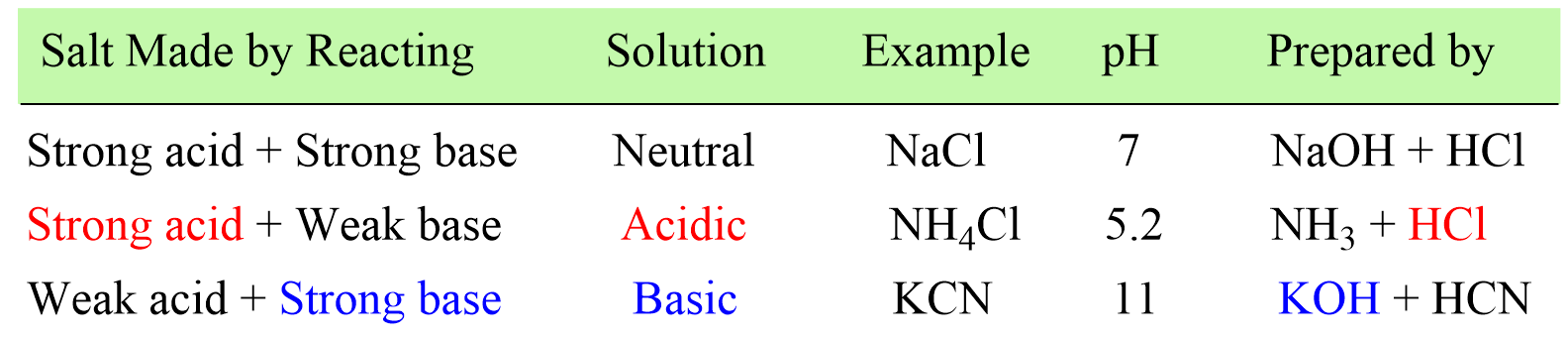
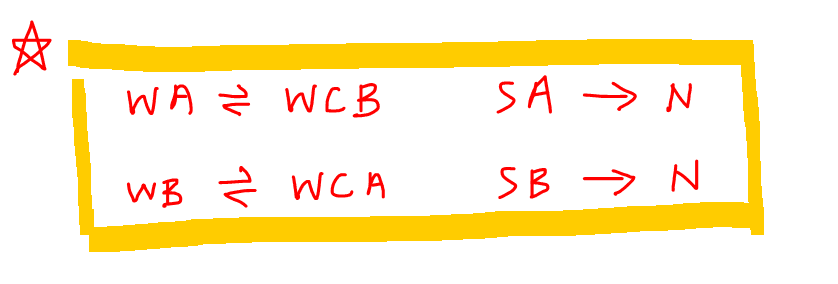
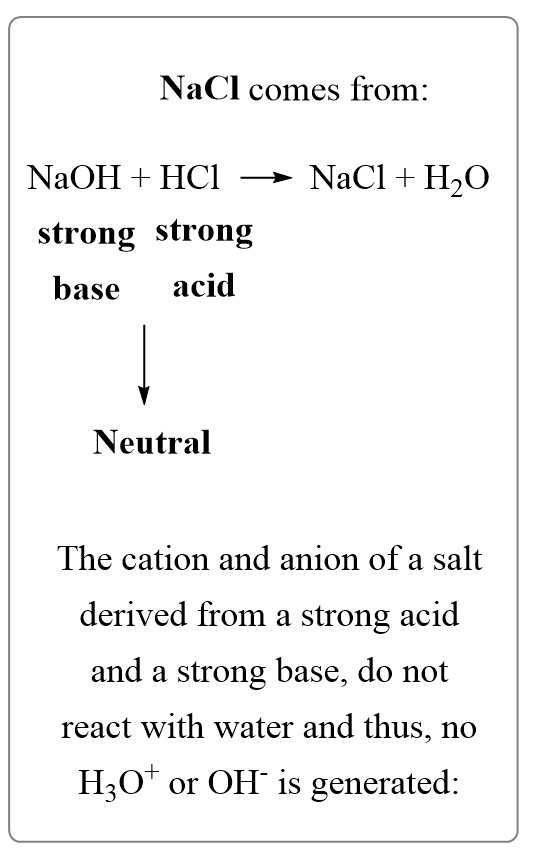
Pure water is netural in nature [H^(+)]=[OH^(-)]. When this condition is disturbed by changing the concentration of H^(+)or OH^(-), the natural solution changes to acidic {[H^(+)]gt[OH^(-)]} or basic {[H^(+)]lt[OH^(-)]}. This change occurs
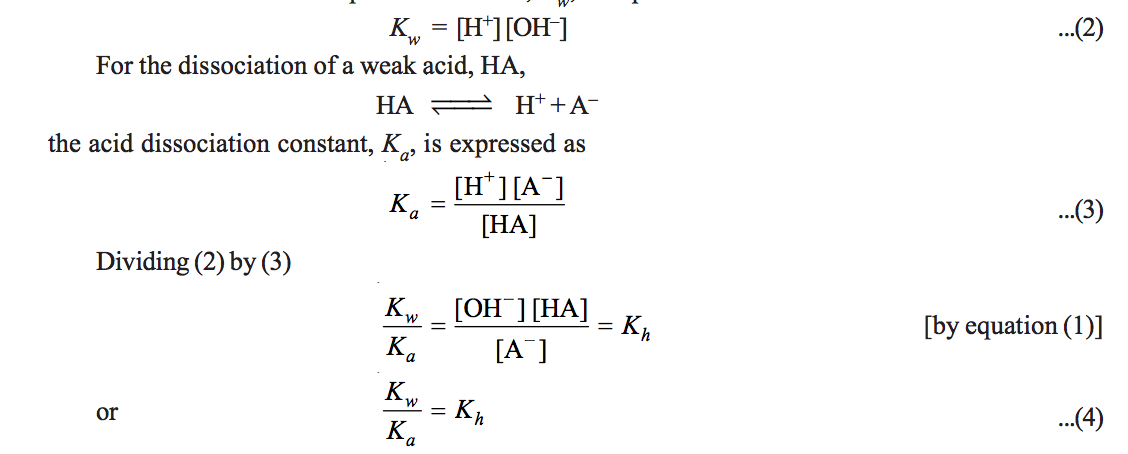
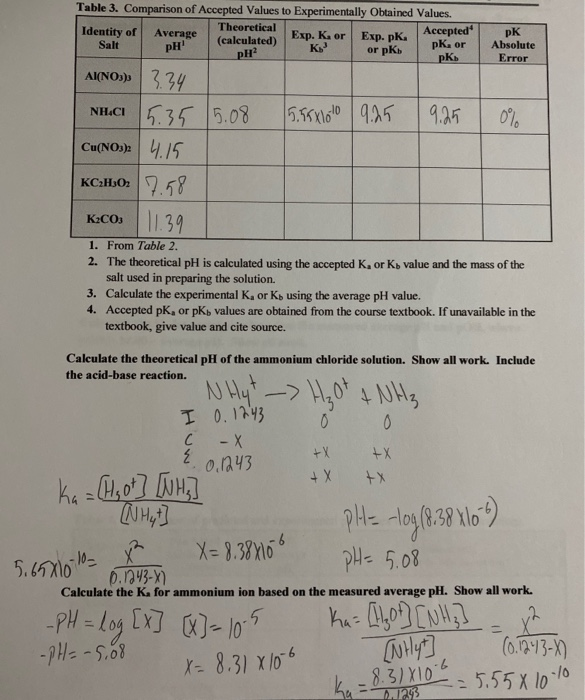
Calculation of Hydrolysis Constant, Degree of Hydrolysis and pH of Salt Solution - Chemistry, Class 11, Ionic Equilibrium

Calculate pH of a solution of given mixture (0.1 mole CH3COOH + 0.2 mole CH3COONa ) in 100 ml 0f mixture. K = 2 × 10^-5

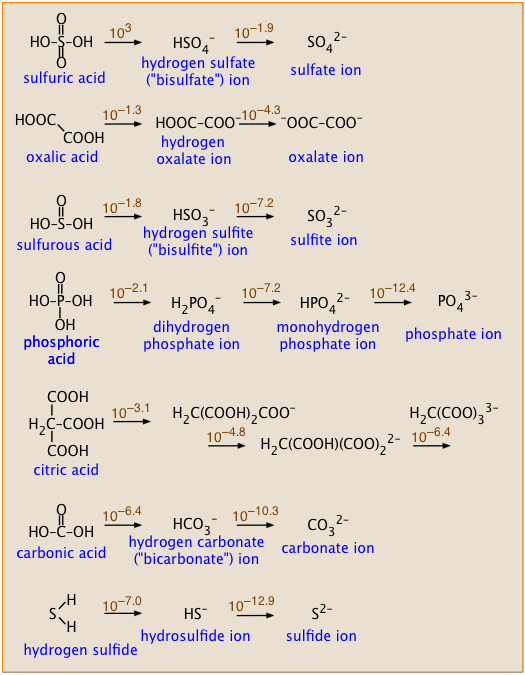
aqueous solution - Accurate method to calculate the pH of a salt from a weak acid and weak base - Chemistry Stack Exchange

Calculate pH of a salt of weak monobasic acid and weak monoacidic base having concentration 0.1 M at 25^oC (Given : - pka = 4.8 pkb = 5.2 )