![Stearic acid [CH3(CH2)16CO2H] is a fatty acid, the part of fat that stores most of the energy. 1.0 g of stearic acid was burned in a bomb calorimeter. The bomb had a Stearic acid [CH3(CH2)16CO2H] is a fatty acid, the part of fat that stores most of the energy. 1.0 g of stearic acid was burned in a bomb calorimeter. The bomb had a](https://dwes9vv9u0550.cloudfront.net/images/3157558/bad535b0-48a3-43b8-b0a2-70b0557725fe.jpg)
Stearic acid [CH3(CH2)16CO2H] is a fatty acid, the part of fat that stores most of the energy. 1.0 g of stearic acid was burned in a bomb calorimeter. The bomb had a

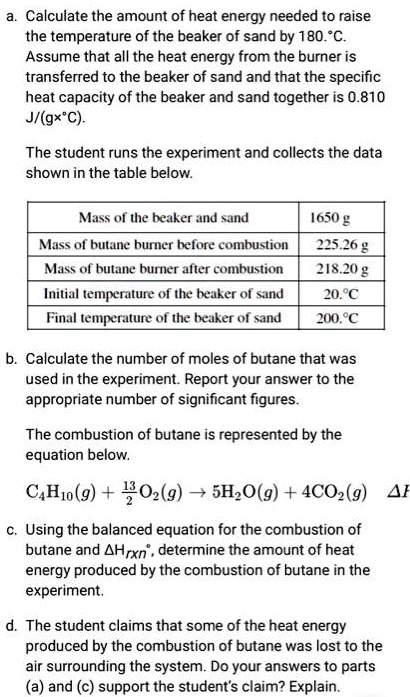
SOLVED: Butane Burner a. Calculate the amount of heat energy needed to raise the temperature of the beaker of sand by 180^∘C. Assume that all the heat energy from the burner is