SciELO - Brasil - Terbinafine: optimization of a LC method for quantitative analysis in pharmaceutical formulations and its application for a tablet dissolution test Terbinafine: optimization of a LC method for quantitative

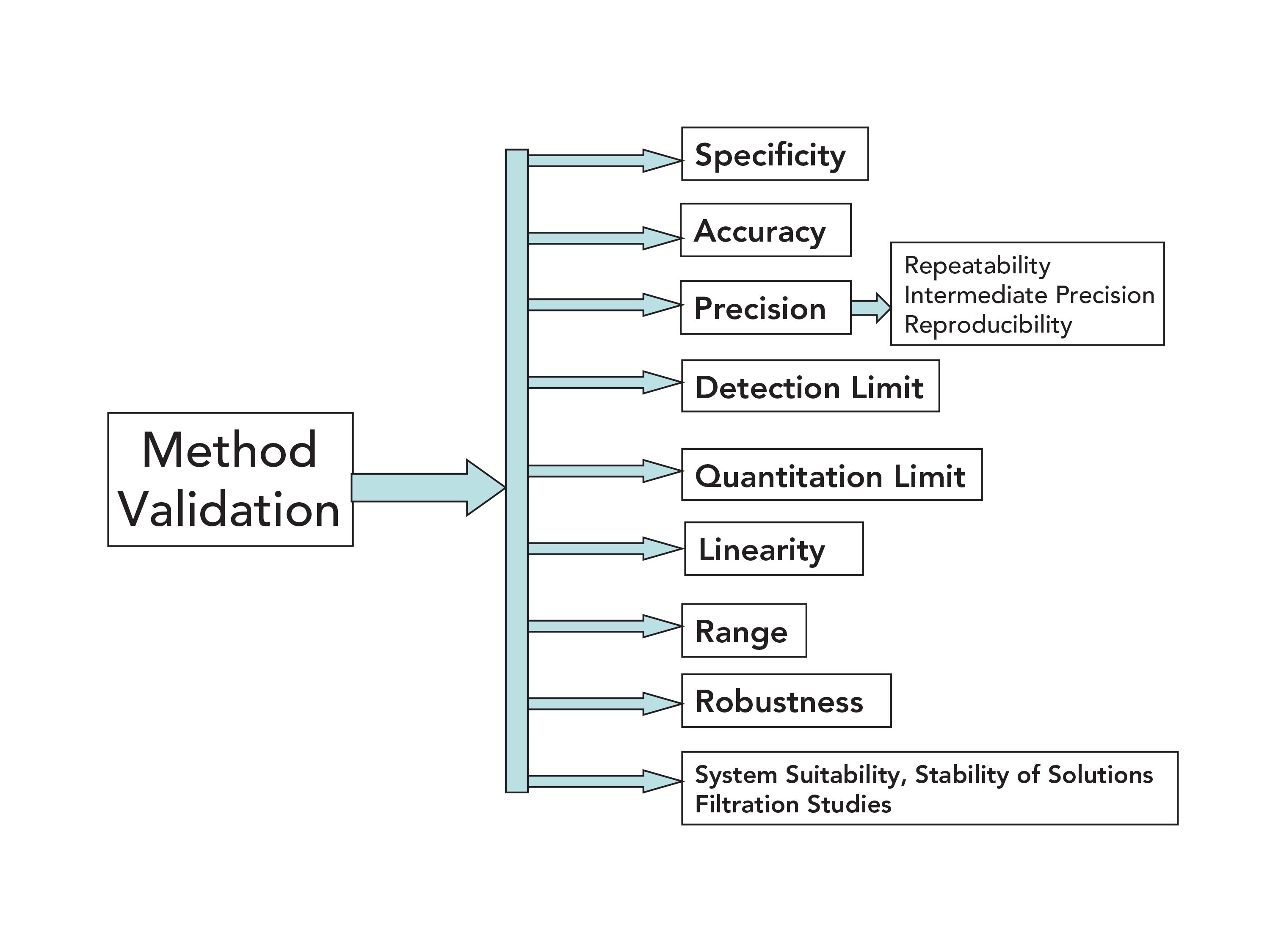
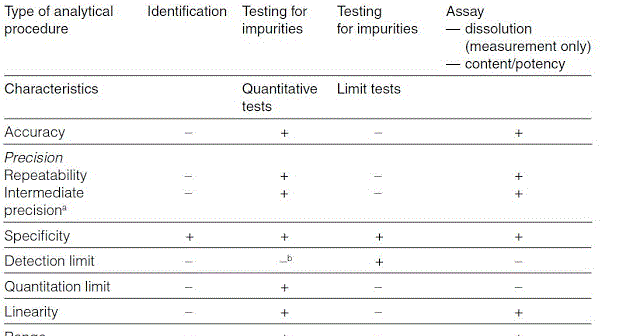
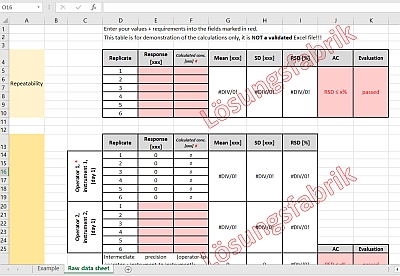
Method Validation Approaches for Pharmaceutical Assessments – Highlights with High Performance Thin Layer Chromatographic (HPTLC) Techniques | IntechOpen

Development and Validation of New RP-HPLC Method for the Estimation of Alfuzosin Hydrochloride in Bulk and Tablet Dosage Form

Analytical Method Validation for Quality Assurance and Process Validation Professionals | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
METHOD VALIDATION BY MEANS OF THE ACCURACY PROFILE: A REALISTIC APPROACH FOR ROUTINE CHEMICAL LABORATORIES

A practical guide to analytical method validation, including measurement uncertainty and accuracy profiles - ScienceDirect